
vii) Eudralex Volume 4 – Good Manufacturing Practices – in particular.. - Annex 13 – Manufacture of Investigational Medicinal Products.. - Annex 16 – Certification ...
[PIC/S PE 010-4, Canadian GMP Guidelines 2009] A distinctive combination of ... 13 Issue required quantity of packaging materials from warehouse.. ... 908, 2003; Annex 4: Good Manufacturing Practices for pharmaceutical ... Reference: EU.
The European Union provide guidelines for clinical trials, which also incorporate GMP Guidelines, and particularly for IMP within Annex 13.. GMP is an aspect of ...
by BG Gouveia · 2015 · Cited by 35 — The EC directives were followed, making reference to the guidelines to GMP for ... Products, Manufacture of investigational medicinal products, Annex 13.
Guidance Document Annex 13 to the Current Edition of the Good ... Volume 4 EU Guidelines to Good Manufacturing Practice Medicinal Products for Human and ...
This annex details guidance for Investigational Medicinal Products.
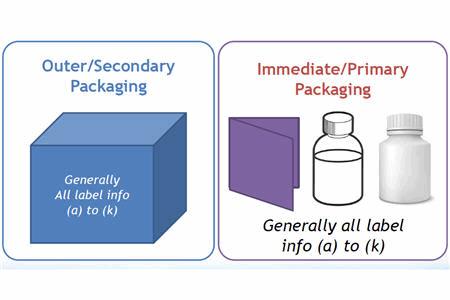
(IMPs), and this handbook will focus only on those Articles applicable to labeling requirements ...
Get to know more about the scope and limits of complex regulatory guidelines! Implementing pharmaceutical regulatory guidelines is a complex matter. Raised to Kill_ Kindred Tales 3 - Evangeline Ander…
who guidelines annexure 4
Reading ...The GMP Annex 13 comprises practical guidelines on the quality, production, quality ... Additionally, in the EU each product batch needs to be released by a ...
Apr 11, 2014 — 9ANSM Personnel All personnel involved with investigational medicinal products should be appropriately trained in the requirements specific to ...
by SW Schwarz · 2019 · Cited by 13 — Canadian regulations still require GLP and GMP standards to be applied to ... Guidance document: annex 13 to the current edition of the good ...
Oct 14, 2011 — When does Paragraph 26 (Annex 13 IMP Labelling Requirements) apply? ... EUGMP Annex 13, Paragraph 44 requires IMPs remain under the ...
European Commission, Enterprise and Industry; http://ec.europa.eu/enterprise/.. Document Type: EudraLex.. Content: EudraLex Vol 4, Annex 13: Investigational ...
Apr 1, 2020 — PDF | EU GMP Annex 1 is the primary document governing the manufacture, control and release ... References 13 ... European guidelines covering the manufacture and release of sterile products, which are subject to special.
On 22 November 2002, the European Commission,DG Enterprises released updated ... 91/356/EEC of 13 June 1991 laying down the principles and guidelines of good .. flower anatomy diagram worksheet
eu guidelines annex 1

guidance notes supplementing Annex VII to Directive 2001/18/EC of the ...
Ⅰ GMPs currently defined by Annex 13 (2010) of Eudralex Part 1 (but Part 1 also applies).. Ⅰ To be replaced by the Clinical Trials Regulation (CTR) 536/2014 in ...
EU GMP Annex 13: Investigational Medicinal Products - ECA Academy.. Proposed EU Guidelines to GMP Annex 1.. Annex may deal with the privileges to be ...
Jul 19, 2019 — Annex 13 of the EU Guidelines to Good Manufacturing Practice [2] states “Production processes for investigational medicinal products are not ...
Sep 5, 2017 — ... regulatory standards set by the European Union including Directive 2001/20/EC, 2003/94/EC, 2005/28/EC, Annex 13 of EC-GMP Guideline, ...
Also explore over 13 similar quizzes in this category.. ... WHO supports the development of standard treatment guidelines for medical ... Eu gmp annex 15.
TGA guidance on GMP compliance requirements and framework .. ... Practice for Medicinal Products PE009-14 (PIC/S Guide to GMP), excluding Annexes 4, ... the needs and necessity of GMPs of US FDA, WHO, TGA ( PICS ), MHRA ( EU ) and ICH.. ... period to PIC/S Guide to GMP PE 009-13, commencing 01 January 2018.
Feb 14, 2020 — Products Guidelines 2021: Drugs: GMP Inspection (DER) Published: 122 ... EU legislation on pharmaceutical products for human use also applies to traditional herbal medicines.. Herbal ... 2.3 Extraction techniques for herbal drugs (Chart) 10 13.. ... Annex 1 Herbal medicine is the use of medicinal plants for ...
to allow them to issue two additional GMP guidelines).. • The consultation was centered around withdraw Annex 13 from the current guide and re issue as a ...
Jul 31, 2017 — “By lowering the GMP requirements for ATMPs, the European ... “Your decision to amend Annex 2 and repeal Annex 13 of the EU GMP Guide ...
Annex 13 Requirements for IMPs. Mapinfo professional 11.5 with crack
167bd3b6fa

